Breakthrough Bandage Heals Wounds Faster Than Ever
No matter what type of wounds need to be healed, the treatments are very similar. They’re typically cleaned with antiseptics and then wrapped in gauze bandages to be shielded from infectious germs, minimize pain, and keep slightly damp.

In the past few years, more refined methods have been introduced to treat blisters, burns, cuts, splinters, scrapes, and punctures. These new methods track people’s healing processes by monitoring temperature and pH, but due to complicated manufacturing processes, customization challenges, and cost, they aren’t really used extensively. What’s more, none of them aggressively assist in the progression of healing.
An Exciting Breakthrough

Researchers at the Wyss Institute for Biologically Inspired Engineering at Harvard University, the Harvard John A. Paulson School for Engineering and Applied Sciences (SEAS), and McGill University recently developed a method that speeds up the healing of wounds — and it could potentially revolutionize treatment methods.
AADs (active adhesive dressings) are based on hydrogels that are heat-sensitive and have all the desirable attributes of wound healing environments. That is, AADs are highly adhesive, mechanically active, antimicrobial, tough, and stretchy. This means they can heal wounds much faster than current methods and stave off infection without any additional tools or medicines.

David Mooney, a founding core faculty member of the Wyss Institute and the Robert P. Pinkas Family Professor of Bioengineering at SEAS, excitedly reports: “This technology has the potential to be used not only for skin injuries, but also for chronic wounds like diabetic ulcers and pressure sores, for drug delivery, and as components of soft robotics-based therapies.”
Embryonic Inspiration
The skin on developing embryos heals itself without developing any scar tissue. The skin cells surrounding embryonic wounds produce actin, a protein in the fiber that contracts to flawlessly close lesions. This characteristic slowly disappears as the embryo matures, and at a certain point, injuries to it start to cause irritation and scarring as they would to any other person.
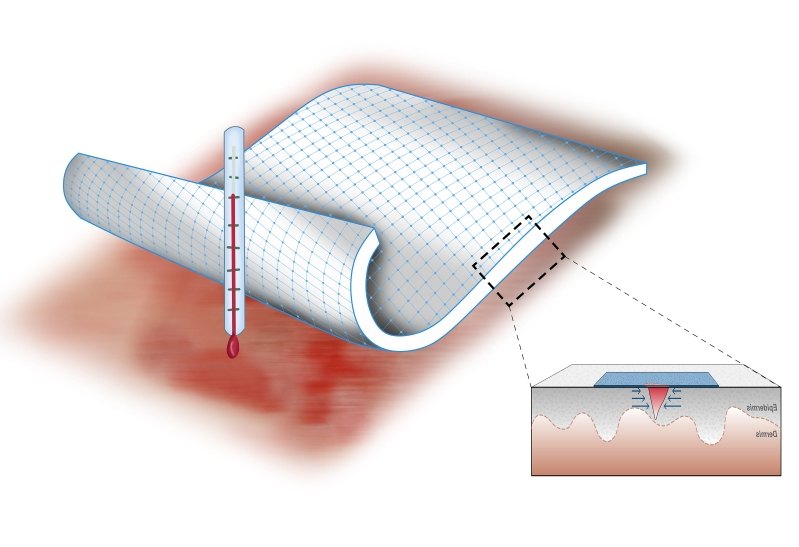
Using this knowledge, researchers enhanced the hydrogels with a thermoresponsive polymer called PNIPAm. Since PNIPAm contracts at about 90 degree Fahrenheit and is water repellent, body heat both repels water and serves to shrink it. The resulting hydrogel conveys the force of the shrinking PNIPAm elements to the primary tissue by forming sturdy links between the bandage and the tissue. On top of that, silver nanoparticles rooted in the AAD offer up antimicrobial shields.
The Pig Skin Test

Since pigs and humans share so many genetic properties, they were the first species to be exposed to the AADs. According to Benjamin Freedman, a Graduate School of Arts and Sciences’ postdoctoral fellow in the Mooney lab who serves as Project Leader, “The AAD bonded to pig skin with over 10 times the adhesive force of a Band-Aid and prevented bacteria from growing, so this technology is already significantly better than most commonly-used wound protection products, even before considering its wound-closing properties.”
Freedman and his partners continue their research with confidence. “We hope to pursue additional preclinical studies to demonstrate AAD’s potential as a medical product, and then work toward commercialization.” Stay tuned for more information on this medical phenomenon.