Purdue University Gets Closer to Quick Coronavirus Testing With Portable Paper Device
In the past few weeks, the CDC’s distribution of test kits for the novel coronavirus (COVID-19) has been limited at best. NPR has reported that only a favored few laboratories across the United States (numbering less than 100 in total) are currently authorized to use the federally mandated test to speed up identification of the virus in patients. But an ever-rising number of cases of COVID-19 has prompted urgent need for better, faster diagnostic tests.
The latest publication of the American Chemical Society’s ACS Omega journal offers some valuable help via a possible innovation from Purdue University. There, a team of researchers led by Biomedical Engineering Professor Jacqueline Linnes has recently devised a diagnostic tool small enough to fit in one’s hand to test for coronavirus.

So far, the prototype device is only capable of detecting the MERS-CoV coronavirus strain. But its detection capability is both accurate and sensitive — even for incredibly small concentrations of the virus. Naturally, this provides hope for creating a similar diagnostic test for COVID-19. The challenge, though, will be in how to manufacture the COVID-19-specific detector not just at the university laboratory scale, but on one large enough to meet all people’s needs in the midst of the current global pandemic.
Linnes’ research has focused on designing portable devices that can quickly diagnose a range of various diseases, especially infectious and virulent ones, like whooping cough and cholera. Conventional diagnostic devices often require lengthy timespans (a few weeks to a couple of months) before results are known. But Linnes has sought to cut down on that time with small paper-based gadgets made from glass fiber or cellulose.
As Purdue University has documented, “paper-based devices [have made] for faster diagnostics, because paper-like materials, such as glass fiber and cellulose, are robust and known to function as a pump…..carry[ing] samples, such as blood or water, without all the external equipment required within a lab….. [and] reading a paper strip is easy and low cost.”
Linnes’ diagnostic tools are distinctive for their efficiency. For instance, they can indicate whether a test sample is positive for a disease in much the same fashion as a pregnancy test. These fast-acting devices can even be reliably used in the comfort of one’s home, with results available in under an hour rather than the typical lengthy weeks’ time.
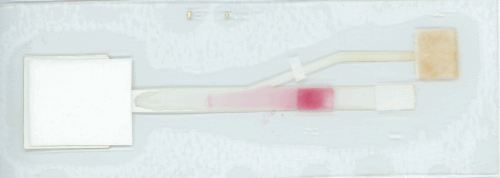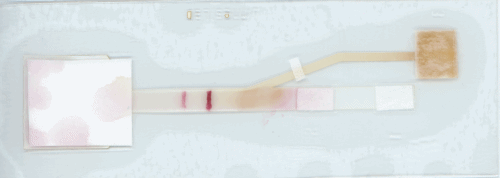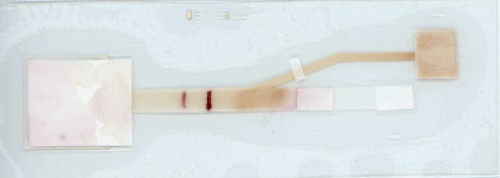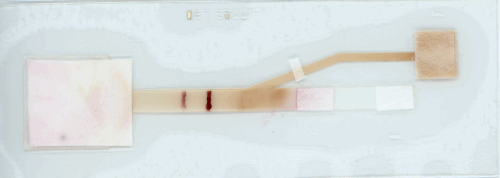
Linnes adds that “because this device has a more complex shape, a process hasn’t been developed to make it available on a commercial scale. However, many processes in electronics and paper manufacturing could be translated to scaling up this device.”
“This paper device isn’t dependent on a particular virus or sequence. To detect COVID-19, we would just need an assay design specific to that sequence, which could come from a nasal or throat swap sample. Just like with MERS-CoV, a user could load the assay with liquid into the paper platform, fold the device, and let it run.”
Linnes’ MERS-CoV diagnostic invention has already been filed for intellectual property (IP) patent protection through Purdue University’s Office of Technology Commercialization (OTC). In other words, this MERS-CoV diagnostic tool is available for license. The various types of licensing agreements Purdue University’s OTC has are outlined at this link here.
Through this licensing process, Linnes’ prototype device can be modified to meet a broader set of needs. As described on ACS Omega, the diagnostic tool “has shown versatility in the ability to detect multiple types of pathogens…this proof-of-concept…shows promise for detecting many other pathogens beyond MERS-CoV,” and with any luck, can swiftly be cross-applied to COVID-19.