FDA Approves Portable 5-Minute Coronavirus Test by Abbott Laboratories
American healthcare and medical device company Abbott Laboratories has just unveiled a portable test for the novel coronavirus. Not only is it unique for being the size of a mere toaster, but even more significantly, it can deliver results in as little as five minutes. Because of the state of emergency that the United States is facing in light of the pandemic, the United States Food and Drug Administration (FDA) has issued an emergency use authorization (EUA) to have the new test available for hospitals, urgent care clinics, and physicians’ offices right away.

The US has struggled to supply hospitals and other patient-care facilities with an adequate number of tests since the beginning of the COVID-19 outbreak. As little as one month ago, testing was restricted to high-risk travelers who had recently returned from journeys to and from China.
Unfortunately, too many people were allowed to spread the disease back then, making the situation much worse and forcing healthcare facilities to start testing a much larger chunk of the public. And even though calls for testing expanded, the pivot created a major logistical challenge. As US Surgeon General Jerome Adams explained on Fox News, “it’s important for people to know…..the CDC was never designed to provide hundreds of millions of tests. It was designed to respond to outbreaks.”

Interestingly, The Atlantic highlighted another key factor affecting the detection of COVID-19, that of the “new coronavirus-testing crisis. Its main cause is not the federal government, nor state public-health labs, but the private companies that now dominate the country’s testing capacity. Testing backlogs have ballooned, slowing efficient patient care and delivering a heavily lagged view of the outbreak to decision makers.”
Dr. Tedros Adhanom Ghebreyesus, the Director-General of the World Health Organization (WHO), similarly urges, “We have a simple message for all countries: test, test, test. All countries should be able to test all suspected cases. They cannot fight this pandemic blindfolded; they should know where the cases are.”
COVID-19’s appearance has upended the status quo of the healthcare landscape, requiring numerous changes in protocol. One of the measures passed to speed things along through the crisis was the invocation of emergency use authorization (EUA), detailed on the FDA’s website. On top of that, section 564 of the Federal Food, Drug, and Cosmetic Act (FD&C Act) permits the FDA Commissioner to “allow unapproved medical products or unapproved uses of approved medical products to be used in an emergency to diagnose, treat, or prevent serious or life-threatening diseases or conditions caused by CBRN [chemical, biological, radiological, and nuclear] threat agents when there are no adequate, approved, and available alternatives.”
As Dr. Anthony Fauci, director for the National Institute of Allergy and Infectious Diseases and member of the White House coronavirus task force, stated to NPR, “in the beginning, it was a slow start. But right now that the commercial firms have gotten involved, we really have caught up. And we will be — we’re seeing a much more improved system with regard to the availability and the implementation of testing.”

Abbott explains that their new test “runs on Abbott’s ID NOW TM platform—a lightweight box (6.6 pounds and the size of a small toaster) that can sit in a variety of locations,” adding that “because of its small size, it can be used in more non-traditional places where people can have their results in a matter of minutes, bringing an alternate testing technology to combat the novel coronavirus. We’re ramping up production to deliver 50,000 ID NOW COVID-19 tests per day, beginning next week, to the U.S. healthcare system.”
Quickly identifying COVID-19 cases helps immensely in the administration of quick treatment, and it’s the only real way to curb the spread of the disease sooner rather than later. In fact, according to the American Heart Association, testing of any communicable disease is of paramount concern. Why? “It’s crucial, of course, to help treat, isolate, or hospitalize people who are infected. Testing also is important in the bigger public health picture on mitigation efforts, helping investigators characterize the prevalence, spread, and contagiousness of the disease.”
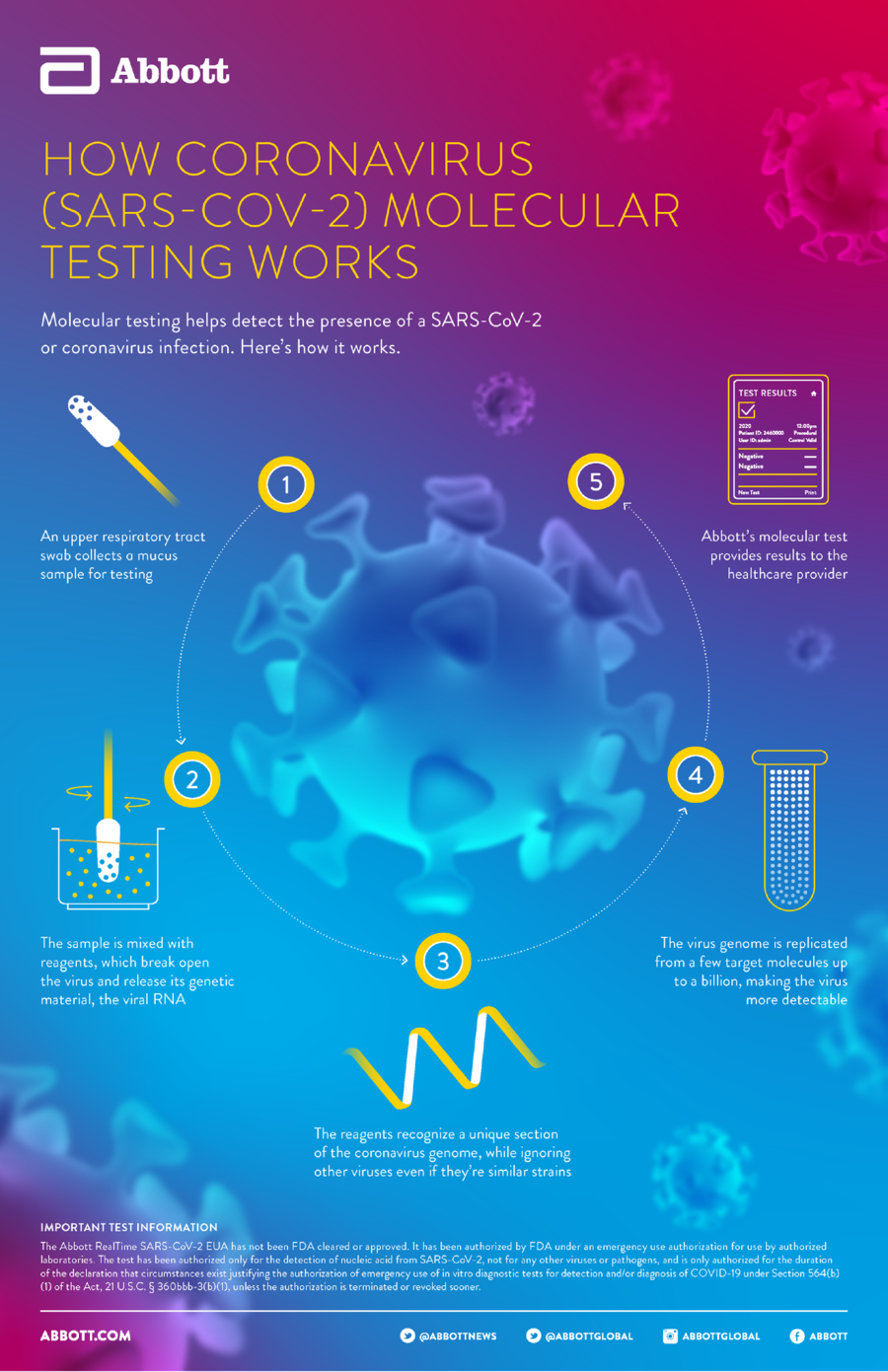
How does the Abbott Laboratories portable detector work? First, a sample specimen is drawn from a patient. Abbott Laboratories has not disclosed on its website what type of specimen is drawn, but it’s believed to come via swabs from the nasal passages or back of the throat, or even respiratory specimens, as the CDC says. The drawn sample is then fed into the detector, where a chemical is combined with it to determine the genetic material of any viruses present and compare them to those found in a large database. If any fragments of the novel coronavirus’s genome are present, the test will yield a positive result in about five to 13 minutes.
With this faster testing system now in play, mitigation measures can be hastened to contain the pandemic. Not only will lives be saved, but the data from all these tests will better inform both public health authorities and government officials of COVID-19’s prevalence and the ways it’s evolving.